You will have a short session on this in the face to face part of the course but it is worth including a brief summary here.

Prevention is better than cure
Rule 1 – Don’t spill anything.
Rule 2 – When you do – spill as little as possible.
So decant into smaller containers, carry carefully (in a carrier for large bottles), place away from edges of benches etc.
Keep yourself safe.
There are some spillages that you should simply leave to the experts:
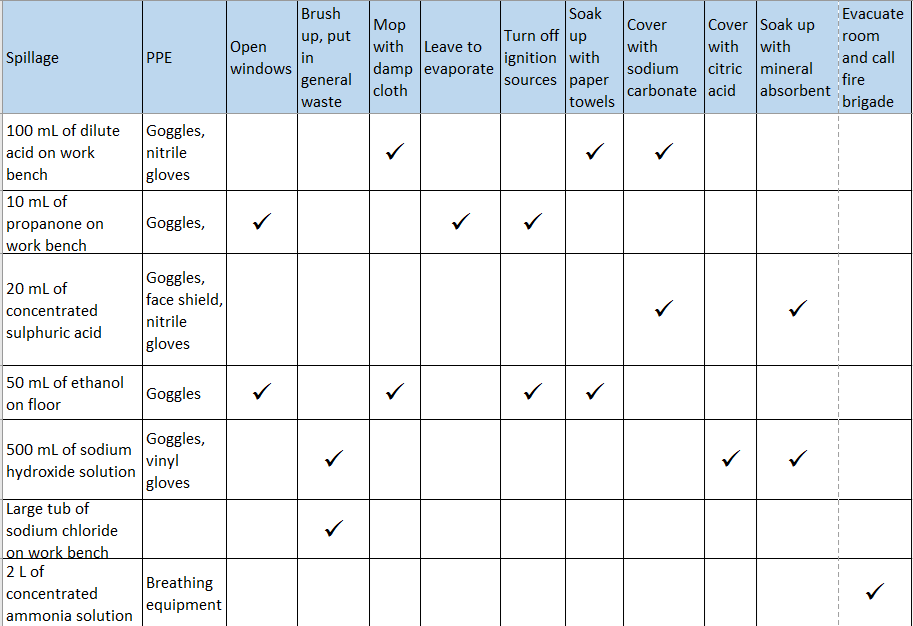
Any large spill that generates hazardous fumes: eg ammonia, bromine, concentrated ethanoic, hydrochloric or nitric acids, any mixture that produces chlorine, nitrogen dioxide or sulphur dioxide etc.


Solids & Liquids
Sweep up in a dustpan and brush and dispose as recommended. Be wary of raising dust.
Liquids can be absorbed into a solid – mineral based cat litter is goodt for this. It can then be swept up and disposed of
Acids and Alkalis
For spills of acids – such as concentrated H2SO4 neutralise with anhydrous sodium carbonate or hydrogencarbonate and then dispose normally
For alkalis such as sodium hydroxide – if a solid, just brush with a dustpan and brush and dispose normally. If a solution, neutralise with citric acid and then scrape up and dispose.


Oils etc
Absorb into cat-litter and clear up as before.
Use detergent to clear up the remnants and make sure the area is safe.
The arrangement documents give some examples in a table. A version of that is here.