Radioactive Decay
The alchemists of days gone by spent a lot of time trying to turn lead into gold. We now know that they could never have succeeded. An element is determined by the number of protons in its nucleus and that is something that cannot be changed by heating it, reacting it with other chemicals and so forth. Carbon is carbon because its nucleus contains 6 protons. If its nucleus had 7 protons, it would be nitrogen. Nuclei also contain neutrons (there is an exception to this… do you know of a nucleus with no neutrons?)
That’s not to say that a nucleus of one element never changes into the nucleus of another. This can happen for certain nuclei. These nuclei are described as ‘unstable’. They can become more stable by transforming into another nucleus and when they do this, radiation is emitted. This process is called radioactive decay.
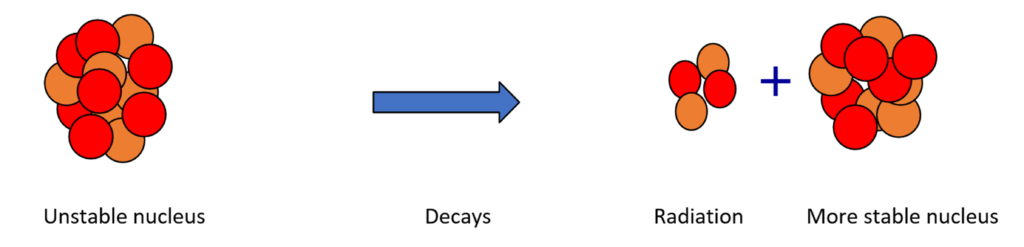
There are three decay mechanisms.
Alpha decay – the unstable nucleus emits an alpha particle. An alpha particle consists of two protons and two neutrons, the same as a helium nucleus.
Beta decay – the unstable nucleus emits a high-speed electron. You might well ask what an electron is doing in the nucleus. It happens because a neutron can transform into a proton and an electron.
Gamma decay– the unstable nucleus has too much energy and loses some in the form of a gamma photon. A gamma photon is like a high energy X-ray. Some people talk about gamma rays rather than gamma photons.
The decay mechanism depends on the nucleus. Here are some examples:
- Americium-241 decays to neptunium-237 by emitting an alpha particle.
- Strontium-90 decays to yttrium-90 by emitting a beta particle.
- Caesium-137 decays to barium-137 by emitting a beta particle. The barium-137 nucleus then emits a gamma photon.
Substances like the ones mentioned above are said to be radioactive. They emit radiation without any stimulus – no heating, electric current or similar. What is more, there is no way to stop them being radioactive (except, perhaps, to wait, sometimes for thousands of years!).
Many nuclei formed by radioactive decay are still unstable and go on to decay themselves, forming a decay chain or decay series.

