While quite a lot of chemicals such as sodium chloride (table salt) or sucrose (sugar) can just go down the sink or into the bin. There are numerous substances used in schools and colleges that could cause damage to the environment. These need to be disposed of properly.
There are two main approaches for those that can’t just go to waste: treat or recycle inhouse or keep for uplift by a licensed contractor.

What to dispose of
Chemicals past their Safe Shelf Life – eg potassium metal.
Chemicals past their Shelf life eg calcium oxide
Excess stock – perhaps bought for AH projects years ago.
Waste from experiments – probably the most regular source of waste.
Down the sink
Anything can go down the sink if it is dilute enough – but it is an offence to dilute something specifically to dispose of it.
Some, need to be treated first. Acids and alkalis, for instance need to be neutralised.

Washings
Washings of anything are OK. By this term we mean what is left after you have emptied the container. You don’t need to try to squeeze every last drop out but even the remains of lead and mercury compounds, once the contnts have been poured out can be washed away.

Organic waste
Where possible, use substances that can simply be washed to waste like ethanol or propanone.
If you can’t, keep quantities to a minimum as it will have to be stored for uplift.
Waste can be stored in mixed waste bottles but these should only be used in the prep room by technicians and you should keep a record of what goes in them.
Organic waste
You can mix different organic wastes BUT halogenated waste, such as dichloromethane, must be kept separate from non-halogenated waste.
Image by Jeremy Brooks on Flickr under a Creative Commons license.


Inoprganic waste
If possible use compoubnds of iron, calcium magnesium sodium or potassiun as most of these can be washed to waste.
If not, reduce the scale where possible. Solutions can be precipitated and filtered as the solid takes much less space – eg add sodium carbonate to lead nitrate solution and filter off the lead carbonate.
Otherwise, compounds of copper, zinc, lead, nickel, chromium and many more need to be kept for uplift.
Where to store waste
In the chemical store, in the same way as if it was not waste eg flammable waste in a flammable cabinet.
It shouldn’t be kept for an excessive period. Ideally it should be disposaed of annually though it is very unlikely any regulator would kick up a fuss if it were kept for longer.

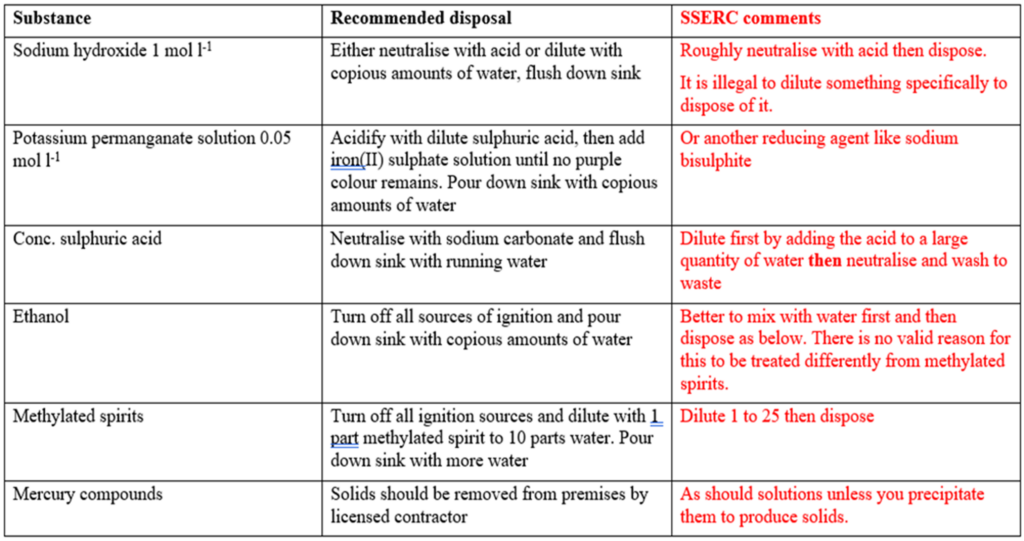
Some disposal examples
Below is a table taken from the support documents with examples of how to dispose of some chemicals – with an extra column containing SSERC comments.